The embryos were good. The transfers were done correctly. And each time, your embryo didn’t make it.
If you have been through this more than once, you already know that recurrent implantation failure is one of the hardest places to be in fertility treatment — because it explains almost nothing about why. Something is preventing implantation. The question of what is rarely answered clearly.
When it isn’t, the immune system is frequently proposed as the reason. And steroids — prednisolone or dexamethasone, prescribed to modulate immune activity around the time of transfer — are among the most commonly offered next steps.
The Human Fertilisation and Embryology Authority, the UK’s independent fertility regulator, is one of the few public bodies anywhere in the world that formally assesses the add-ons offered alongside standard IVF. Their evidence reviews are used internationally by clinicians and patients trying to understand what the research actually shows.
Steroids for recurrent implantation failure are on that list.
What the immune theory is based on
Implantation requires the maternal immune system to do something counterintuitive. It must partially suppress itself — tolerating an embryo that carries paternal antigens it would ordinarily identify as foreign.
The theory behind immune add-ons is that this balance is harder to achieve in some women. The immune environment remains more active than implantation requires. Steroids, as anti-inflammatory and immune-modulating agents, are offered to support that shift.
The reasoning has a biological basis. The question the HFEA addresses is whether intervening in that signalling produces more live births.
What the regulator assessment shows
The HFEA reviews each add-on against the evidence for whether it improves the chance of a live birth.
The evidence for steroids in recurrent implantation failure is weak. Routine use across the general population of women with implantation failure is not currently supported.
The clinical situations where the case for steroids may be stronger are specific:
- A diagnosed autoimmune condition — rheumatoid arthritis, lupus, thyroid autoimmunity — where immune dysregulation is already documented
- Endometriosis, where chronic peritoneal inflammation is part of the clinical picture
- Elevated inflammatory markers identified in blood tests that a consultant is specifically responding to
- A reproductive immunologist has reviewed the full picture and identified a specific immune pattern worth addressing
If your recommendation for steroids came with that level of clinical rationale behind it — a named condition, a specific test result, a specialist assessment — the conversation is a different one. The regulator’s assessment covers the broader population. Your situation may sit within the narrower group where the evidence is more supportive.
If steroids were offered as a general next step after failed transfers, without that specific clinical picture behind them, the assessment is useful information to bring into the conversation with your consultant about what the reasoning is and whether it applies to you.
The regulator’s work is designed to inform that conversation, not replace it.
Where your influence sits
Waiting isn’t neutral
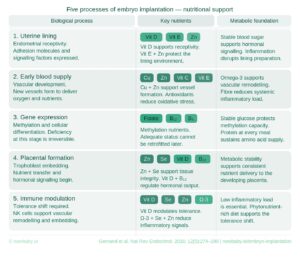
Implantation unfolds in stages.
Each stage responds to the nutritional and metabolic environment around it. These are the variables that remain within your influence.
More than 1 in 3 frozen transfers do not result in a live birth. The physiological environment during the two-week wait is within your capacity to support.
From the moment of transfer, your body is working.
The Now Baby FET Implantation Meal Plan was built around every one of them. Professionally analysed. Beginning the day after transfer.
You didn’t come this far to wing it.