You are ready to transfer. The embryo is stored. But your lining is not where your clinic wants it to be — not thick enough, not responding the way it should, not yet creating the conditions the embryo needs to arrive into.
Platelet-rich plasma, infused directly into the uterine cavity, is one of the options now being offered in this situation. PRP — prepared from your own blood — delivers concentrated growth factors to the endometrium with the aim of supporting lining development and improving receptivity before transfer.
The Human Fertilisation and Embryology Authority, the UK’s independent fertility regulator, formally assesses the add-ons offered alongside standard IVF. Their evidence reviews are used internationally by clinicians and patients trying to understand what the research actually shows.
Intrauterine PRP is on that list.
What intrauterine PRP involves
A sample of your own blood is drawn and spun in a centrifuge to concentrate the platelets. The resulting plasma — rich in growth factors involved in tissue repair and regeneration — is infused into the uterine cavity, typically in the cycle before or the cycle of transfer.
The biological rationale is that growth factors delivered directly to the endometrium may support lining thickness, improve receptivity and promote the adhesion molecules and signalling factors that allow an embryo to attach and embed.
What the regulator assessment shows
The HFEA reviews each add-on against the evidence for whether it improves the chance of a live birth.
On intrauterine PRP, the evidence base is promising but not yet sufficient to support routine use. Small studies have reported improvements in endometrial thickness and implantation rates, particularly in women with persistently thin lining or recurrent implantation failure. Larger randomised controlled trials are ongoing. The HFEA currently rates the evidence as insufficient — the signal is there, but the quality of evidence needed to recommend it routinely is not yet in place.
The clinical situations where intrauterine PRP may be considered are specific:
- A persistently thin endometrial lining resistant to standard oestrogen preparation
- Asherman’s syndrome or intrauterine adhesions affecting lining development
- Recurrent implantation failure where lining receptivity has been identified as a contributing factor
If your recommendation for intrauterine PRP came with that specific clinical picture behind it, the conversation with your consultant is a different one. If it was offered without that rationale, the assessment is useful information to bring into that conversation.
The regulator’s work is designed to inform that conversation, not replace it.
What the lining needs to do this
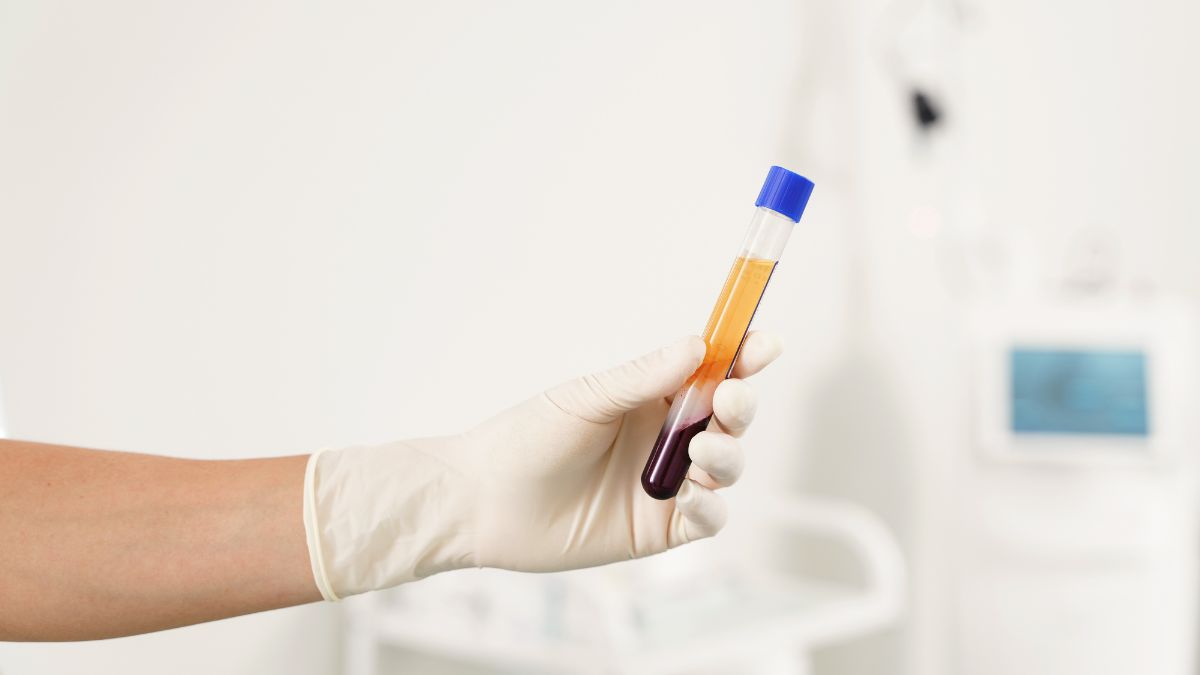
PRP stimulates the endometrium — but it cannot fuel what it stimulates. What the lining is built from, day by day across the cycle, is determined by the nutritional environment the body is carrying into that preparation. This is a gap that you can fill.
Vitamin A supports the development of the uterine lining and the adhesion factors that allow implantation to begin. Zinc contributes to tissue formation and the signalling processes the endometrium depends on. Iron supports the blood supply that a thickening lining requires. These are the inputs PRP has to work with — and the same inputs implantation depends on when transfer comes.
The next milestone is implantation.
The two-week wait is not neutral — it is biologically active. Embedding, vascular development, placental formation, immune adaptation — each stage has its own specific nutrient requirements..
The Now Baby FET Implantation Meal Plan was built for targeted implantation support. Professionally analysed. Beginning the day after transfer.
You didn’t come this far to wing it.